 alkali, and alkaline-earth metals, tend to be good reducing agents, as their valence electrons, whose radial orbit DEFINES the atomic radius, tend to be readily oxidized. On the other hand, the larger elements, i.e. The atomic radius of a chemical element Oxygen Nitrogen Carbon. nitrogen, fluorine, oxygen, TEND to be very powerful oxidants, and this is also manifested by their small atomic size. The general trend is that atomic sizes increase as one moves downwards in the Periodic Table of the Elements, as electrons fill outer electron shells. Atomic Radius - trends on periodic table Generally, the atomic radius decreases across a period from left to right and increases down a given group. Periodic trends of atomic radius, relative sizes of element atoms, vector diagram, atomic.  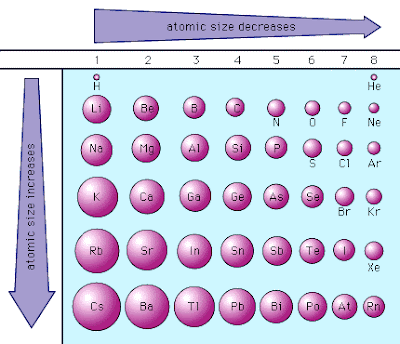
Excluding the Noble Gases, the smaller atoms from the right hand side, i.e. It follows that the SMALLEST atoms derive the right of the Table as we face it. Of course, the diagram shows NO data (it should do so), but the relative size of the atoms across the Period, and down the Group is clear. And the best metric that illustrates this trend is the well-known diminution of atomic radii across the Period from left to right? And of course, we should look at some data. Going down the group, the first ionisation. Within a period, protons are added to the nucleus as electrons are being added to the same principal energy level. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. The atomic radius increases as you move down a column because for every new row of the table a new electron shell is added to the atom. The atomic radius of atoms generally decreases from left to right across a period. The atomic radius for atoms of an element tends to go up as you move down a group of elements in the table. 
Now it is a fact that the nuclear charge is SHIELDED very poorly by incomplete electronic shells. Progressing down group 2, the atomic radius increases due to the extra shell of electrons for each element. While the atomic radius can be defined in a number of different ways, the general atomic radius trend across the periodic table holds true. The chemistry and atomic structure of the elements is a contest between (i) nuclear charge, conveniently represented by #Z_"the atomic number"#, and (ii) shielding by other electrons. #"Increase in atomic radii down a Group, a column of the Periodic"#"Table."# 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
